Kv7.4
Description: potassium voltage-gated channel, KQT-like subfamily, member 4 Gene: Kcnq4 Alias: KV7.4, DFNA2, DFNA2A, KCNQ4
Kv7.4 (also known as DFNA2 or DFNA2A), encoded by KCNQ4, is a members of the potassium voltage-gated channel KQT-like subfamily.
Kv7.4 It is thought to play a critical role in the regulation of neuronal excitability, particularly in sensory cells of the cochlea. Defects in this gene are a cause of nonsyndromic sensorineural deafness type 2 (DFNA2), an autosomal dominant form of progressive hearing loss. NCBI
Experimental data
Rat Kv7.4 gene in CHO host cells |
||
|
Click for details 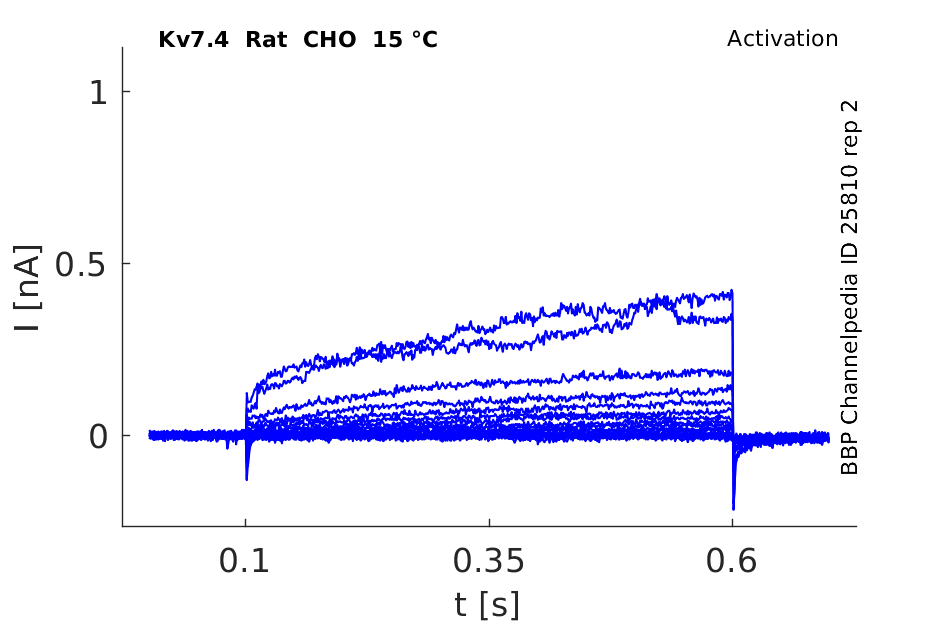
15 °Cshow 71 cells |
Click for details 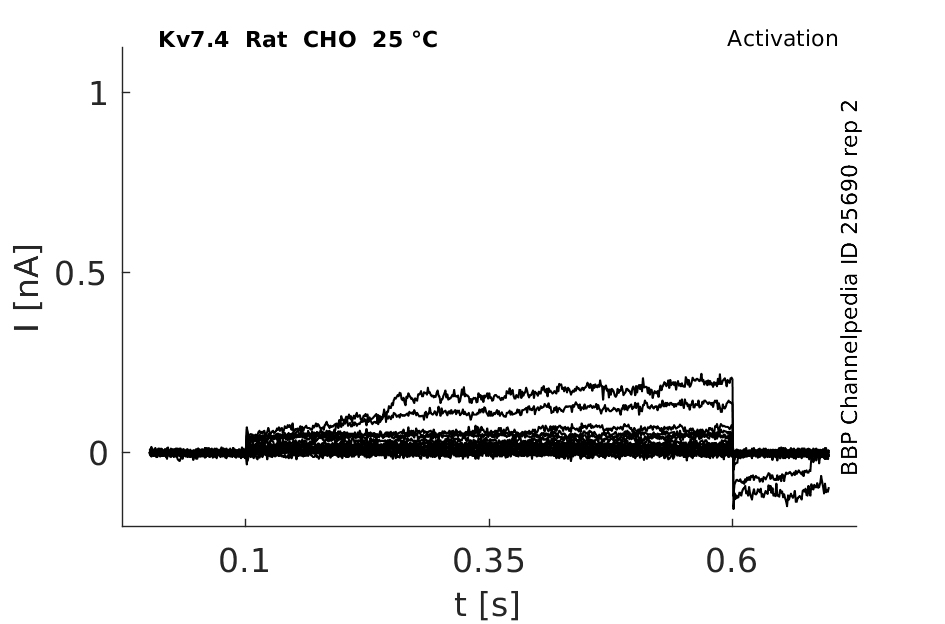
25 °Cshow 106 cells |
Click for details 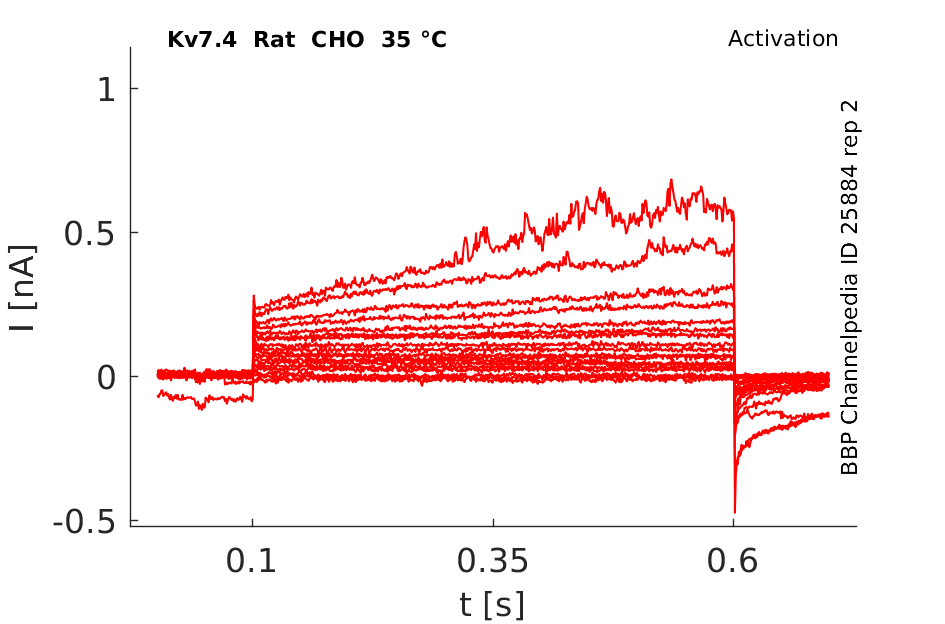
35 °Cshow 121 cells |
Two transcript variants encoding different isoforms have been found for this gene (see Protein Isoform)
| Species | NCBI accession | Length (nt) | |
|---|---|---|---|
| Human | NM_004700.4 | 4324 | |
| Mouse | NM_001081142.3 | 4070 |
Two transcript variants encoding different isoforms have been found for this gene
Isoforms
Post-Translational Modifications
Visual Representation of Kv7.4 Structure
Methodology for visual representation of structure available here
Structural Model of Kv7.4
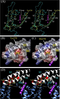
KCNQ4 Structure with DFNA2 MUTATION
KCNQ4 channel belongs to the family of voltage-gated K+ channels, which consists of six transmembrane domains (S1–S6) and a K+ selective pore. Most of missense mutations associated with DFNA2 affect the pore structure of the channels, exerting strong dominant negative effects on the channel function. Deletions, on the other hand, cause a frame-shift, resulting in the truncated channels that are nonfunctional. Clinically, patients with deletions have milder hearing loss than that observed in the patients with missense mutations [11]. Further study of KCNQ4 mutations will improve our understanding of the molecular mechanisms of progressive hearing loss [1684]
Kv7.4 predicted AlphaFold size
Methodology for AlphaFold size prediction and disclaimer are available here
KCNQ4 Kinetics in CHO cells
Using the whole-cell configuration of the patch-clamp technique, we have investigated the characteristics of these transfected cells. The average cell capacitance of the CHO cells was 22.2 ± 8.7 pF (mean ± S.D., n = 21). The outward current showed properties of KCNQ4. The current activated with a time constant of 110 ± 46 ms when the potential was stepped to 0 mV. At 0 mV the current was 2.64 ± 1.46 nA in amplitude. The current was half activated at a potential of -28.8 ± 8.0 mV. In eight cells, outward current was almost completely blocked by 200 µM linopirdine (mean inhibition 82 %). The anti-arrhythmic bepridil, a blocker of KCNQ1, applied at 10 µM, also blocked 49 % (n = 3) of the CHO outward currents (University College London (2003) J Physiol 547P, PC21)
KCNQ4 expressed in CHO cells+ Various Channel Openers
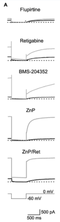
Kv7.4 Expressed in HEK and CHO cells Comparison

Expression in the Brain
While the Kv7.2 and Kv7.3 subunits are present in almost all brain regions examined so far [724] the Kv7.4 subunit is expressed only in discrete nuclei of brainstem, including the mid- brain [762], [761].
KCNQ4 is found only in a few nuclei and tracts mainly in the brainstem [762]
Kv7.4 can be found in substantia nigra pars compacta (SNc) and ventral tegmental area (VTA), [762].
The M type K+ channel, whose molecular basis is considered to be KCNQ2-5, has been characterized in many typesof peripheral and central neurons, including superior cervical ganglion (SCG), dorsal root ganglion (DRG), hippocampal and cortical neurons (Owen et al., 1990 [1088]; Passmore et al., 2003 [1089]; Peretz et al., 2005 [78]; Shah et al., 2002 [700]).
Expression In Body
KCNQ4 current is a low-threshold, non-inactivating K+ current, which is expressed in the outer hair cell of cochlea, brain, heart, and skeletal muscle. (Su [137])
Hair Cells
KCNQ4 can also associate with KCNQ3 and yield M-type currents, but its expression pattern is much more restricted. It is prominently expressed in sensory hair cells in the inner ear8, and in certain tracts and nuclei of the central auditory pathway [464]
KCNQ4 Cellular Distribution in Neuron
Kv7.4 channels are expressed only in mesencephalic dopaminergic neurons at somatodendritic sites (see fig 1 in [752]).
Stablizing membrane potential
The M current plays a key role in regulating various central and peripheral neuron excitabilities and stabilizing membrane potential (Delmas and Brown, 2005 [1090], [1091]).
Deafness
Mutations in KCNQ4 channel produce inherited syndrome of deafness Kubisch ([763]). The deficit of KCNQ4 function might result in a chronic potassium overload of outer hair cells, causing their slow degeneration. (Su [137])
Mutations in KCNQ4 lead to a slowly progressive, dominant hearing loss [464]
KCNQ4 K(+) Channels Tune Mechanoreceptors
Mutations inactivating the potassium channel KCNQ4 (K(v)7.4) lead to deafness in humans and mice. In addition to its expression in mechanosensitive hair cells of the inner ear, KCNQ4 is found in the auditory pathway and in trigeminal nuclei that convey somatosensory information. We have now detected KCNQ4 in the peripheral nerve endings of cutaneous rapidly adapting hair follicle and Meissner corpuscle mechanoreceptors from mice and humans.
Down regulation of Kv7.4 in Hypertension
In 2 different rat and mouse models of hypertension, the functional impact of Kv7 channels was dramatically downregulated.
Modulation of KCNQ4 channel activity by changes in cell volume
KCNQ4 channels expressed in HEK 293 cells are sensitive to cell volume changes, being activated by swelling and inhibited by shrinkage, respectively. The KCNQ4 channels contribute significantly to the regulatory volume decrease (RVD) process following cell swelling. Under isoosmotic conditions, the KCNQ4 channel activity is modulated by protein kinases A and C, G protein activation, and a reduction in the intracellular Ca2+ concentration, but these signalling pathways are not responsible for the increased channel activity during cell swelling [1818]
Kv7.3/Kv7.2
The Kv7.4 subunit coassembles with the Kv7.3, but not the Kv7.2, subunit, which produces larger currents than homomeric Kv7.4 channels per se [763].
KCNE Subunits
Coexpression of the KCNE- Beta-subunits with human KCNQ4 in the Xenopus laevis oocyte expression system revealed that all KCNEs modulate KCNQ4 voltage dependence, protein stability and ion selectivity of hKCNQ4 in Xenopus oocytes. The deafness-associated Jervell and Lange- Nielsen syndrome (JLNS) mutation KCNE1(D76N) impairs KCNQ4-function whereas the Romano-Ward syndrome (RWS) mutant KCNE1(S74L), which shows normal hearing in patients, does not impair KCNQ4 channel function. In conclusion, KCNEs are presumably coexpressed with KCNQ4 in hair cells from the organ of Corti and might regulate KCNQ4 functional properties, effects that could be important under physiological and pathophysiological conditions [1817]
Linopirdine
When Kv7.4 forms a heteromer with Kv7.3, the resulting potassium channel conductance is more sensitive to linopirdine [763]
Retigabine
The principal Kv7 channel opener, retigabine, is able to produces a hyperpolarizing shift of the activation curve of the channel by 14–43 mV (depending on the Kv7 channel subtype) at 10 μm. KCNQ4 channels, stably expressed in HEK293 cells, were activated by retigabine and BMS-204352 in a reversible and concentration-dependent manner in the concentration range 0.1–10 μM. Both compounds shifted the KCNQ4 channel activation curves towards more negative potentials by about 10 mV. Further, the maximal current obtainable at large positive voltages was also increased concentration-dependently by both compounds. (Schroder [136])
Mepyramine
Mepyramine inhibits the individual homomeric KCNQ1-4 channels. (Liu [72])
Ca2+
M currents in mammalian neuron are inhibited but in amphibian are enhanced by [Ca2+]. (Su [137])
Ionomycin
Ionomycin enhances the KCNQ4 current which is expressed in Xenopus oocytes. The enhanced effect is reversed by the application of BAPTA-AM, a fast calcium chelator. Surprisingly, the intracellular injection of calcium (0.01–1 mM) into the cytoplasm directly did not change the KCNQ4 currents. These data demonstrated that the effect of ionomycin acts on intramembrane site of KCNQ4 protein and without relation to cytoplasmic calcium concentration. (Su [137])
ML213 and NS15370
Recently, two novel Kv7 channel enhancers have been identified, ML213 and NS15370, that show increased potency, particularly on Kv7.4 channels. This study identifies and characterises ML213 and NS15370 as potent vasorelaxants in different blood vessels, thereby highlighting these new compounds as potential therapeutics for various smooth muscle disorders.
Janus kinase 2
Janus kinase-2 (JAK2) participates in the signaling of several hormones, growth factors and cytokines. JAK2 downregulates KCNQ4 activity and thus counteracts K(+) exit, an effect which may contribute to cell volume regulation [1812]
BDNF Increases expression of Kv7.4
BDNF profoundly and specifically increases KCNQ4 expression in neurons derived from embryonic stem cells [1813]
References
Liu B
et al.
Antihistamine mepyramine directly inhibits KCNQ/M channel and depolarizes rat superior cervical ganglion neurons.
Neuropharmacology,
2008
Mar
, 54 (629-39).
Peretz A
et al.
Meclofenamic acid and diclofenac, novel templates of KCNQ2/Q3 potassium channel openers, depress cortical neuron activity and exhibit anticonvulsant properties.
Mol. Pharmacol.,
2005
Apr
, 67 (1053-66).
Grunnet M
et al.
hKCNE4 inhibits the hKCNQ1 potassium current without affecting the activation kinetics.
Biochem. Biophys. Res. Commun.,
2005
Mar
25
, 328 (1146-53).
Schrøder RL
et al.
KCNQ4 channel activation by BMS-204352 and retigabine.
Neuropharmacology,
2001
Jun
, 40 (888-98).
Su CC
et al.
Studies of the effect of ionomycin on the KCNQ4 channel expressed in Xenopus oocytes.
Biochem. Biophys. Res. Commun.,
2006
Sep
15
, 348 (295-300).
Jentsch TJ
Neuronal KCNQ potassium channels: physiology and role in disease.
Nat. Rev. Neurosci.,
2000
Oct
, 1 (21-30).
Brown DA
et al.
Muscarinic suppression of a novel voltage-sensitive K+ current in a vertebrate neurone.
Nature,
1980
Feb
14
, 283 (673-6).
Wang HS
et al.
KCNQ2 and KCNQ3 potassium channel subunits: molecular correlates of the M-channel.
Science,
1998
Dec
4
, 282 (1890-3).
Shah MM
et al.
Molecular correlates of the M-current in cultured rat hippocampal neurons.
J. Physiol. (Lond.),
2002
Oct
1
, 544 (29-37).
Pan Z
et al.
A common ankyrin-G-based mechanism retains KCNQ and NaV channels at electrically active domains of the axon.
J. Neurosci.,
2006
Mar
8
, 26 (2599-613).
Jensen HS
et al.
Inactivation as a new regulatory mechanism for neuronal Kv7 channels.
Biophys. J.,
2007
Apr
15
, 92 (2747-56).
Schroeder BC
et al.
KCNQ5, a novel potassium channel broadly expressed in brain, mediates M-type currents.
J. Biol. Chem.,
2000
Aug
4
, 275 (24089-95).
de Heer AM
et al.
Audioprofile-directed successful mutation analysis in a DFNA2/KCNQ4 (p.Leu274His) family.
Ann. Otol. Rhinol. Laryngol.,
2011
Apr
, 120 (243-8).
Hernandez CC
et al.
Regulation of neural KCNQ channels: signalling pathways, structural motifs and functional implications.
J. Physiol. (Lond.),
2008
Apr
1
, 586 (1811-21).
Hansen HH
et al.
Kv7 channels: interaction with dopaminergic and serotonergic neurotransmission in the CNS.
J. Physiol. (Lond.),
2008
Apr
1
, 586 (1823-32).
Wang Q
et al.
[KCNQ4 gene mutations affected a pedigree with autosomal dominant hereditary hearing loss]
Zhonghua Er Bi Yan Hou Ke Za Zhi,
2002
Oct
, 37 (343-7).
Talebizadeh Z
et al.
Novel mutation in the KCNQ4 gene in a large kindred with dominant progressive hearing loss.
Hum. Mutat.,
1999
, 14 (493-501).
Su TR
et al.
Functional study of the effect of phosphatase inhibitors on KCNQ4 channels expressed in Xenopus oocytes.
Acta Pharmacol. Sin.,
2009
Sep
, 30 (1220-6).
Chambard JM
et al.
Regulation of the voltage-gated potassium channel KCNQ4 in the auditory pathway.
Pflugers Arch.,
2005
Apr
, 450 (34-44).
Hansen HH
et al.
The KCNQ channel opener retigabine inhibits the activity of mesencephalic dopaminergic systems of the rat.
J. Pharmacol. Exp. Ther.,
2006
Sep
, 318 (1006-19).
Jones S
et al.
Bradykinin excites rat sympathetic neurons by inhibition of M current through a mechanism involving B2 receptors and G alpha q/11.
Neuron,
1995
Feb
, 14 (399-405).
Zaika O
et al.
Inositol triphosphate-mediated Ca2+ signals direct purinergic P2Y receptor regulation of neuronal ion channels.
J. Neurosci.,
2007
Aug
15
, 27 (8914-26).
Gutman GA
et al.
International Union of Pharmacology. XLI. Compendium of voltage-gated ion channels: potassium channels.
Pharmacol. Rev.,
2003
Dec
, 55 (583-6).
Dalby-Brown W
et al.
K(v)7 channels: function, pharmacology and channel modulators.
,
2006
, 6 (999-1023).
Kharkovets T
et al.
KCNQ4, a K+ channel mutated in a form of dominant deafness, is expressed in the inner ear and the central auditory pathway.
Proc. Natl. Acad. Sci. U.S.A.,
2000
Apr
11
, 97 (4333-8).
Kubisch C
et al.
KCNQ4, a novel potassium channel expressed in sensory outer hair cells, is mutated in dominant deafness.
Cell,
1999
Feb
5
, 96 (437-46).
Tatulian L
et al.
Effect of the KCNQ potassium channel opener retigabine on single KCNQ2/3 channels expressed in CHO cells.
J. Physiol. (Lond.),
2003
May
15
, 549 (57-63).
Li Q
et al.
Identification of novel KCNQ4 openers by a high-throughput fluorescence-based thallium flux assay.
,
2011
Jul
5
, ().
Owen DG
et al.
M-current noise and putative M-channels in cultured rat sympathetic ganglion cells.
J. Physiol. (Lond.),
1990
Dec
, 431 (269-90).
Passmore GM
et al.
KCNQ/M currents in sensory neurons: significance for pain therapy.
J. Neurosci.,
2003
Aug
6
, 23 (7227-36).
Delmas P
et al.
Functional organization of PLC signaling microdomains in neurons.
Trends Neurosci.,
2004
Jan
, 27 (41-7).
Delmas P
et al.
Pathways modulating neural KCNQ/M (Kv7) potassium channels.
Nat. Rev. Neurosci.,
2005
Nov
, 6 (850-62).
Nie L
KCNQ4 mutations associated with nonsyndromic progressive sensorineural hearing loss.
,
2008
Oct
, 16 (441-4).
Jepps TA
et al.
Vasorelaxant effects of novel Kv7.4 channel enhancers ML213 and NS15370.
Br. J. Pharmacol.,
2014
Jun
9
, ().
Leitner MG
et al.
Rescue of ion channel function in deafness-causing KCNQ4 mutants by synthetic channel openers.
,
2011
Sep
26
, ().
Hosseinzadeh Z
et al.
Downregulation of KCNQ4 by Janus kinase 2.
J. Membr. Biol.,
2013
Apr
, 246 (335-41).
Purcell EK
et al.
BDNF profoundly and specifically increases KCNQ4 expression in neurons derived from embryonic stem cells.
Stem Cell Res,
2013
Jan
, 10 (29-35).
Namba K
et al.
In silico modeling of the pore region of a KCNQ4 missense mutant from a patient with hearing loss.
BMC Res Notes,
2012
, 5 (145).
Heidenreich M
et al.
KCNQ4 K(+) channels tune mechanoreceptors for normal touch sensation in mouse and man.
Nat. Neurosci.,
2012
Jan
, 15 (138-45).
Jepps TA
et al.
Downregulation of Kv7.4 channel activity in primary and secondary hypertension.
Circulation,
2011
Aug
2
, 124 (602-11).
Strutz-Seebohm N
et al.
Functional coassembly of KCNQ4 with KCNE-beta- subunits in Xenopus oocytes.
Cell. Physiol. Biochem.,
2006
, 18 (57-66).
Hougaard C
et al.
Modulation of KCNQ4 channel activity by changes in cell volume.
Biochim. Biophys. Acta,
2004
Jan
28
, 1660 (1-6).
Schrøder RL
et al.
Voltage-independent KCNQ4 currents induced by (+/-)BMS-204352.
Pflugers Arch.,
2003
Aug
, 446 (607-16).
Howard RJ
et al.
Structural insight into KCNQ (Kv7) channel assembly and channelopathy.
Neuron,
2007
Mar
1
, 53 (663-75).
Contributors: Rajnish Ranjan, Michael Schartner, Katherine Johnston
To cite this page: [Contributors] Channelpedia https://channelpedia.epfl.ch/wikipages/26/ , accessed on 2026 May 31