Kir2.4
Description: potassium inwardly-rectifying channel, subfamily J, member 14 Gene: Kcnj14 Alias: Kir2.4, IRK4, Kcnj14
The channel kir2.4 encoded by the gene KCNJ14 (also known asIRK4; KIR2.4; KIAA1942; MGC46111) is an integral membrane protein and inward-rectifier type potassium channel, subfamily J, member 14, and probably has a role in controlling the excitability of motor neurons. Two transcript variants encoding the same protein have been found for this gene.
http://www.ncbi.nlm.nih.gov/gene/3770
Strong inwardly rectifying K+ (KIR) channels have been described in many vascular and visceral tissues from different species (Quayle et al., 1997 [941];Nilius & Droogmans, 2001 [942]). These channels pass most current at potentials hyperpolarised to the K+ equilibrium potential (EK), though the small amount of outward carried at voltages depolarised to this potential, is enough to regulate the resting membrane potential (Em) and to cause blood vessel dilatation in response to either raised extracellular (10-15 mM) K+ (Quayle et al., 1997 [941]) or shear stress (Hoger et al., 2002 [943]). Since their initial identification in rat cerebral and coronary artery (Edwards et al., 1988 [944];Quayle et al., 1993 [945];Knot et al., 1996 [946]), patch-clamp studies have reported strongly rectifying inward currents in isolated lung endothelial and bronchial smooth muscle cells (Voets et al., 1996 [947];Kamouchi et al., 1997 [948];Snetkov & Ward, 1999 [949];Michelakis et al., 2001 [950];Hogg et al., 2002 [951];Oonuma et al., 2002 [952];Shimoda et al., 2002 [953]). Regardless of their origin within the cardiovascular system, KIR currents recorded to date are potently blocked by micromolar external Ba2+ ions in a manner that is steeply voltage and time-dependent (Quayle et al., 1993 [945];Robertson et al., 1996 [954];Bradley et al., 1999 [955];Snetkov & Ward, 1999 [949];Sakai et al., 2002 [956];Oonuma et al., 2002 [952]). Summary from [940].
Experimental data
Rat Kir2.4 gene in CHO host cells |
||
|
Click for details 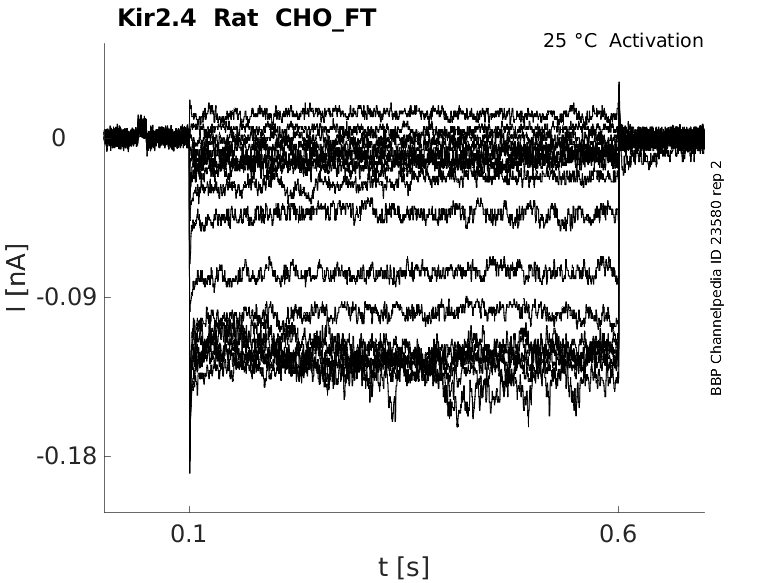
25 °Cshow 45 cells |
Click for details 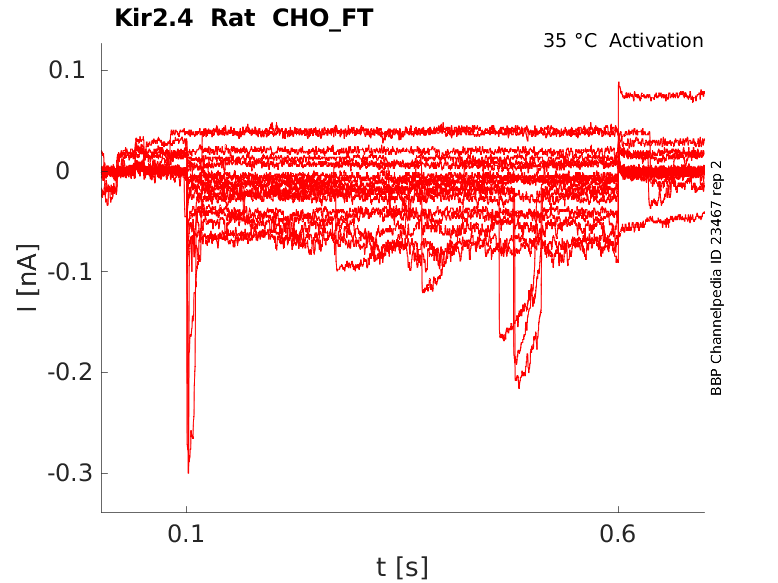
35 °Cshow 21 cells |
|
Kir2.4 (IRK4), which shares 53-63% similarity to Kir2.1, Kir2.2, or Kir2.3 on the amino acid level. Toepert [959]
Transcript
| Species | NCBI accession | Length (nt) | |
|---|---|---|---|
| Human | NM_013348.4 | 3854 | |
| Mouse | NM_145963.2 | 2639 | |
| Rat | NM_170718.2 | 2649 |
Protein Isoforms
Isoforms
Post-Translational Modifications
These channels form as tetramers of subunits that have only two membrane- spanning regions between which is the H5 loop responsible for potassium selectivity. Four isoforms have been identified, Kir2.1, 2.2, 2.3 and 2.4 (Stanfield et al., 2002 [957]).
Kir2.4 predicted AlphaFold size
Methodology for AlphaFold size prediction and disclaimer are available here
Rat Kir2.4 expressed in X.oocytes

Kir2.4 expressed in COS-7 Cells

Single Channel Conductance of Kir2.4
Single-channel recordings revealed strong inwardly rectifying channels with an average conductance of 21 pS in HPASM cells, not significantly different from either Kir2.1 (19.6 pS) or Kir2.4 (19.4 pS). Reverse-transcription polymerase chain reaction detected products corresponding to Kir2.1, Kir2.2 and Kir2.4 but not Kir2.3. We demonstrate that cultured HPASM cells express K(IR) channels and suggest both Kir2.1 and Kir2.4 subunits contribute to these channels, although the whole-cell current characteristics described share more similarity with Kir2.4 [940]
At the molecular level, the Kir2.0 subfamily almost certainly encode the classical inward rectifiers found in the brain, heart, skeletal and vascular muscle (Quayle et al., 1997 [941];Stanfield et al., 2002 [957]). From [940].
Kir2.4 was found in the neural retina. [958]
Expression of Kir2.4 in Rat Brain
In situ hybridization analysis identifies Kir2.4 as the most restricted of all Kir subunits in the brain. Kir2. 4 transcripts are expressed predominantly in motoneurons of cranial nerve motor nuclei within the general somatic and special visceral motor cell column and thus are uniquely related to a functional system. Toepert [959]
X-ray film autoradiographs of sagittal (A) and coronal (B) sections show high mRNA expression in nuclei of the special visceral motor cell column, in the hypoglossal nucleus, and in the choroid plexus [959]
Kir2.4 channels are of major importance in controlling excitability of motoneurons in situ. Toepert [959]
Strong inwardly rectifying K+ (KIR) channels that contribute to maintaining the resting membrane potential are encoded by the Kir2.0 family (Kir2.1-2.4). [940]
Kir2.4 co expressed with Kir2.1

Barrium
Cloned Kir2.4 and KIR currents in HPASM cells showed little voltage dependence to Ba2+ inhibition, which blocked at a more superficial site than for Kir2.1. [940]
Ph
Expression of human Kir2. 4 cRNA in Xenopus oocytes generated strong, inwardly rectifying K(+) currents that were enhanced by extracellular alkalinization. Human Kir2.4 encodes an inwardly rectifying K(+) channel that is preferentially expressed in the neural retina and that is sensitive to physiological changes in extracellular pH. [958]
Ba2+ andCs+
Similar to other Kir2 channels, Kir2.4 is susceptible to block by the extracellular cations Ba2+ and Cs+, but with considerable differences in affinity. The voltage dependence of the Cs+ occlusion between −80 mV and −150 mV membrane potential may indicate Cs+ binding at deeper sites in the open pore where it crossed part of the membrane electric field. Quantitative analysis demonstrated that a tenfold change in K i (i.e., the concentration of Cs+ producing 50% block) corresponded to a change in membrane potential of 36 mV similar to other Kir2 channels [959]
References
Schram G
et al.
Kir2.4 and Kir2.1 K(+) channel subunits co-assemble: a potential new contributor to inward rectifier current heterogeneity.
J. Physiol. (Lond.),
2002
Oct
15
, 544 (337-49).
Tennant BP
et al.
Functional expression of inward rectifier potassium channels in cultured human pulmonary smooth muscle cells: evidence for a major role of Kir2.4 subunits.
J. Membr. Biol.,
2006
, 213 (19-29).
Quayle JM
et al.
ATP-sensitive and inwardly rectifying potassium channels in smooth muscle.
Physiol. Rev.,
1997
Oct
, 77 (1165-232).
Nilius B
et al.
Ion channels and their functional role in vascular endothelium.
Physiol. Rev.,
2001
Oct
, 81 (1415-59).
Hoger JH
et al.
Shear stress regulates the endothelial Kir2.1 ion channel.
Proc. Natl. Acad. Sci. U.S.A.,
2002
May
28
, 99 (7780-5).
Edwards FR
et al.
Inward rectification in rat cerebral arterioles; involvement of potassium ions in autoregulation.
J. Physiol. (Lond.),
1988
Oct
, 404 (455-66).
Quayle JM
et al.
Inward rectifier K+ currents in smooth muscle cells from rat resistance-sized cerebral arteries.
Am. J. Physiol.,
1993
Nov
, 265 (C1363-70).
Knot HJ
et al.
Extracellular K(+)-induced hyperpolarizations and dilatations of rat coronary and cerebral arteries involve inward rectifier K(+) channels.
J. Physiol. (Lond.),
1996
Apr
15
, 492 ( Pt 2) (419-30).
Voets T
et al.
Membrane currents and the resting membrane potential in cultured bovine pulmonary artery endothelial cells.
J. Physiol. (Lond.),
1996
Nov
15
, 497 ( Pt 1) (95-107).
Kamouchi M
et al.
Modulation of inwardly rectifying potassium channels in cultured bovine pulmonary artery endothelial cells.
J. Physiol. (Lond.),
1997
Nov
1
, 504 ( Pt 3) (545-56).
Snetkov VA
et al.
Ion currents in smooth muscle cells from human small bronchioles: presence of an inward rectifier K+ current and three types of large conductance K+ channel.
Exp. Physiol.,
1999
Sep
, 84 (835-846).
Michelakis ED
et al.
Potassium channels regulate tone in rat pulmonary veins.
Am. J. Physiol. Lung Cell Mol. Physiol.,
2001
Jun
, 280 (L1138-47).
Hogg DS
et al.
Endothelial cells freshly isolated from small pulmonary arteries of the rat possess multiple distinct k+ current profiles.
Lung,
2002
, 180 (203-14).
Oonuma H
et al.
Inward rectifier K(+) current in human bronchial smooth muscle cells: inhibition with antisense oligonucleotides targeted to Kir2.1 mRNA.
Am. J. Respir. Cell Mol. Biol.,
2002
Mar
, 26 (371-9).
Shimoda LA
et al.
Inhibition of inwardly rectifying K(+) channels by cGMP in pulmonary vascular endothelial cells.
Am. J. Physiol. Lung Cell Mol. Physiol.,
2002
Aug
, 283 (L297-304).
Robertson BE
et al.
Inward rectifier K+ currents in smooth muscle cells from rat coronary arteries: block by Mg2+, Ca2+, and Ba2+.
Am. J. Physiol.,
1996
Aug
, 271 (H696-705).
Bradley KK
et al.
Kir2.1 encodes the inward rectifier potassium channel in rat arterial smooth muscle cells.
J. Physiol. (Lond.),
1999
Mar
15
, 515 ( Pt 3) (639-51).
Sakai H
et al.
Molecular and pharmacological properties of inwardly rectifying K+ channels of human lung cancer cells.
Eur. J. Pharmacol.,
2002
Jan
25
, 435 (125-33).
Stanfield PR
et al.
Constitutively active and G-protein coupled inward rectifier K+ channels: Kir2.0 and Kir3.0.
Rev. Physiol. Biochem. Pharmacol.,
2002
, 145 (47-179).
Hughes BA
et al.
Cloning and functional expression of human retinal kir2.4, a pH-sensitive inwardly rectifying K(+) channel.
Am. J. Physiol., Cell Physiol.,
2000
Sep
, 279 (C771-84).
Töpert C
et al.
Kir2.4: a novel K+ inward rectifier channel associated with motoneurons of cranial nerve nuclei.
J. Neurosci.,
1998
Jun
1
, 18 (4096-105).
Töpert C
et al.
Cloning, structure and assignment to chromosome 19q13 of the human Kir2.4 inwardly rectifying potassium channel gene (KCNJ14).
Mamm. Genome,
2000
Mar
, 11 (247-9).
Credits
To cite this page: [Contributors] Channelpedia https://channelpedia.epfl.ch/wikipages/45/ , accessed on 2024 May 08