Kv6.1
Description: potassium voltage-gated channel, subfamily G, member 1 Gene: Kcng1 Alias: Kv6.1, kcng1, kh2, kcng, k13
Kv6.1, encoded by the gene KCNG1, is a member is a gamma subunit of the voltage-gated potassium channel, subfamily G. Kv6.1 is thought to be a delayed-rectifier type channels that may contribute to cardiac action potential repolarization. NCBI
Experimental data
Rat Kv6.1 gene in CHO host cells datasheet |
||
|
Click for details 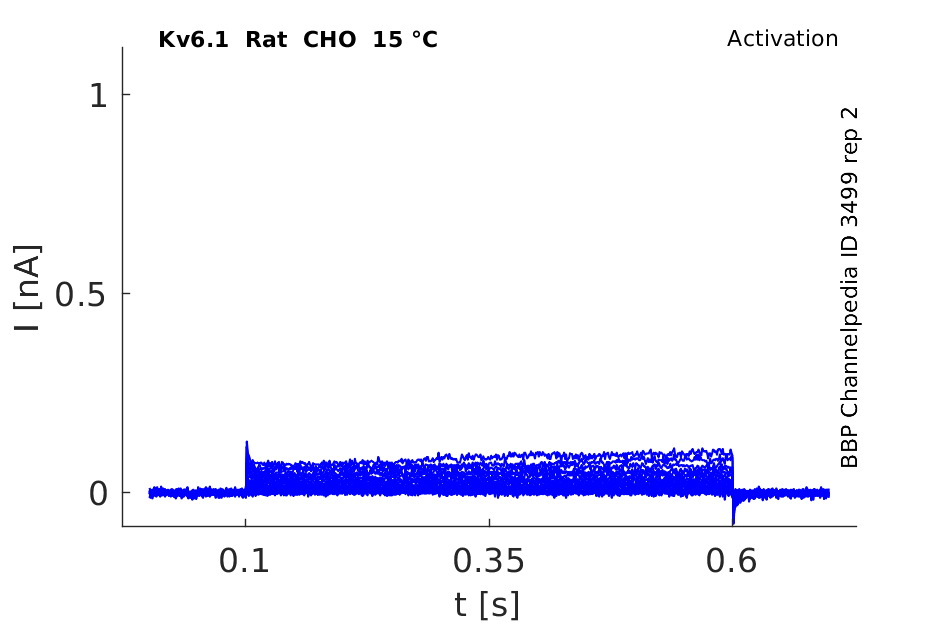
15 °Cshow 41 cells |
Click for details 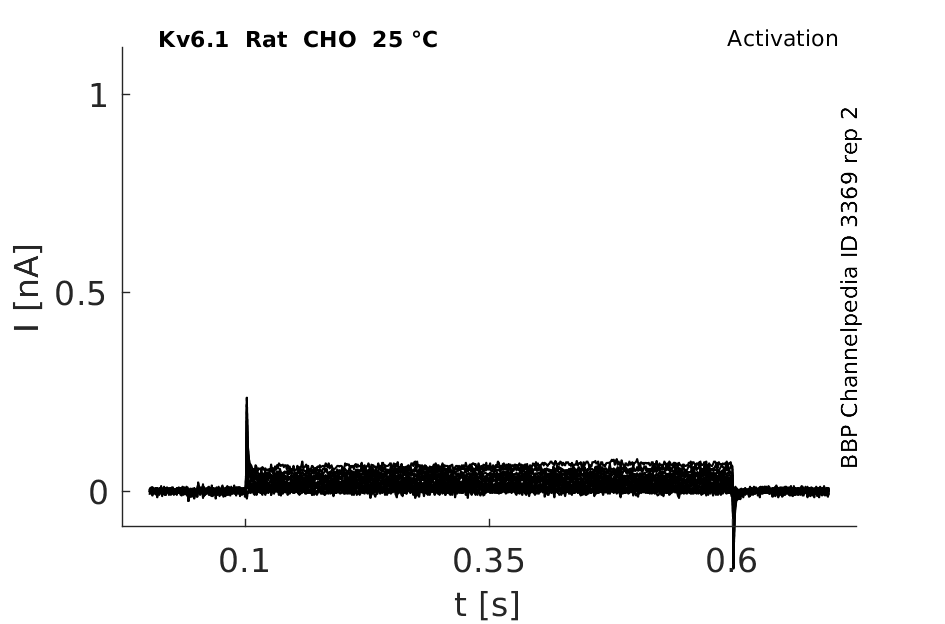
25 °Cshow 92 cells |
Click for details 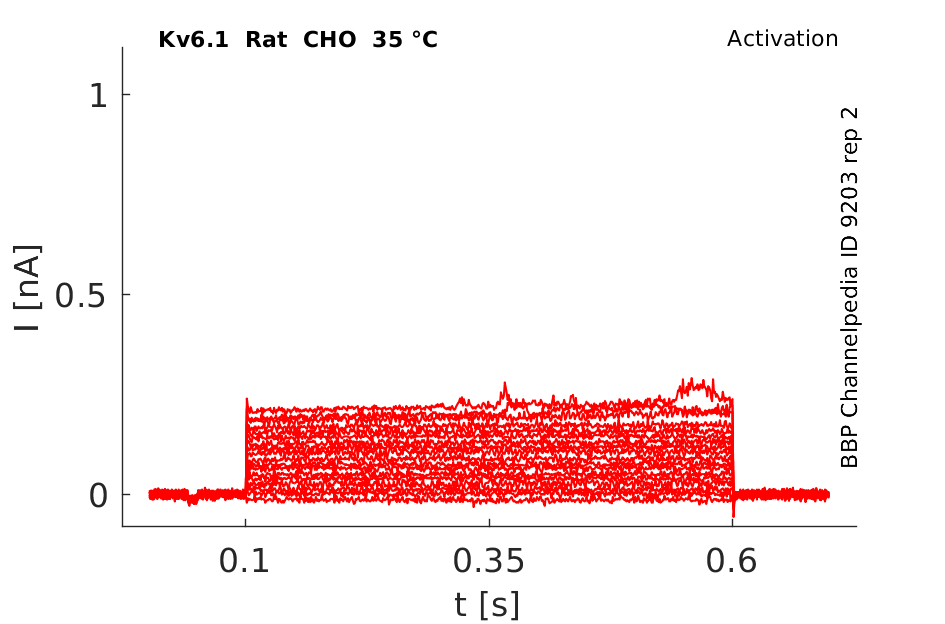
35 °Cshow 69 cells |
Gene
Transcript
| Species | NCBI accession | Length (nt) | |
|---|---|---|---|
| Human | NM_002237.4 | 2189 | |
| Mouse | NM_001081134.2 | 4063 | |
| Rat | NM_001106545.1 | 2745 |
Protein Isoforms
Isoforms
Post-Translational Modifications
Visual Representation of Kv6.1 Structure
Methodology for visual representation of structure available here
Comparisons of predicted amino acid sequences strongly indicate that both Kv5.1 and Kv6.1 are members of the Kv family [399], exhibiting hallmarks such as the conserved GYGD sequence in H5 [657], six hydrophobic transmembrane domains including the positively charged S4 [658],[659], and amino terminal Tl [8] or NA and Nn [399],[660] domains.
Kv6.1 predicted AlphaFold size
Methodology for AlphaFold size prediction and disclaimer are available here
Heteromultimeric potassium channels may include alpha-subunits, such as Kv6.1, that are electrically silent when expressed alone, as is the case for the Kir2 subfamily [652], cyclic nucleotide gated channels [653], [654], [655] and perhaps the Kv4 subfamily [656].
KV6.1 Kinetics with Kv2
We previously showed that coexpression of Kv2.1 and Kv6.1 resulted in currents that deactivated extremely slowly upon depolarization [398]
Human Kv6.1/ Kv6.3/ Kv6.4 Expressed with human Kv2.1 in CHO-K1 Cells

EXPRESSION OF KV6.1
Unlike kH1, 2.4 kb of kH2 was expressed predominantly in the brain, placenta, and the skeletal muscle where it shared a differently spliced form of the kH2 mRNA, approximately 2.0 kb [1698]
Expression in heart
All three known members of the Kv4 family are expressed in the ferret heart, with Kv4.2 being the most abundant (P≤.01). The transcript of Kv5.1 was most common in the right atrium (46.4%) and rarest in the atrial septum (21.5%). Kv6.1 was less abundant. It was present in 28.4% of SA nodal cells and in <16% of cells in the other anatomic regions [1774]
Kv6.1 regulates the kinetics of Kv2.2 channels: It was much less effective in speeding inactivation at intermediate potentials than Kv5.1, had a slowing effect on inactivation at strong depolarizations, and had no effect on cumulative inactivation. Kv6.1 had profound effects on activation, including a negative shift of the steadystate activation curve and marked slowing of deactivation tail currents. [389]
Regulation of gating by electrically silent alpha-subunits, such as Kv6.1, is not restricted to the Kv2 subfamily. They interact with members of the Shal [651] and Kv3 (Shaw) [400] subfamilies as well.
Amino terminal portions of Kv6.1 were unable to form homomultimers but interacted specifically with amino termini of Kv2.1. Xenopus oocytes co-injected with Kv6.1 and Kv2.1 cRNAs exhibited a novel current with decreased rates of deactivation, decreased sensitivity to TEA block, and a hyperpolarizing shift of the half maximal activation potential when compared to Kv2.1. Our results indicate that Kv channel subfamilies can form heteromultimeric channels and, for the first time, suggest a possible functional role for the Kv6 subfamily.
References
Rettig J
et al.
Inactivation properties of voltage-gated K+ channels altered by presence of beta-subunit.
Nature,
1994
May
26
, 369 (289-94).
Kramer JW
et al.
Modulation of potassium channel gating by coexpression of Kv2.1 with regulatory Kv5.1 or Kv6.1 alpha-subunits.
Am. J. Physiol.,
1998
Jun
, 274 (C1501-10).
Post MA
et al.
Kv2.1 and electrically silent Kv6.1 potassium channel subunits combine and express a novel current.
FEBS Lett.,
1996
Dec
9
, 399 (177-82).
Salinas M
et al.
New modulatory alpha subunits for mammalian Shab K+ channels.
J. Biol. Chem.,
1997
Sep
26
, 272 (24371-9).
Isacoff EY
et al.
Evidence for the formation of heteromultimeric potassium channels in Xenopus oocytes.
Nature,
1990
Jun
7
, 345 (530-4).
Jegla T
et al.
A novel subunit for shal K+ channels radically alters activation and inactivation.
J. Neurosci.,
1997
Jan
1
, 17 (32-44).
Namba N
et al.
Kir2.2v: a possible negative regulator of the inwardly rectifying K+ channel Kir2.2.
FEBS Lett.,
1996
May
20
, 386 (211-4).
Chen TY
et al.
A new subunit of the cyclic nucleotide-gated cation channel in retinal rods.
Nature,
1993
Apr
22
, 362 (764-7).
Liman ER
et al.
A second subunit of the olfactory cyclic nucleotide-gated channel confers high sensitivity to cAMP.
Neuron,
1994
Sep
, 13 (611-21).
Bradley J
et al.
Heteromeric olfactory cyclic nucleotide-gated channels: a subunit that confers increased sensitivity to cAMP.
Proc. Natl. Acad. Sci. U.S.A.,
1994
Sep
13
, 91 (8890-4).
Biel M
et al.
Molecular cloning and expression of the Modulatory subunit of the cyclic nucleotide-gated cation channel.
J. Biol. Chem.,
1996
Mar
15
, 271 (6349-55).
Haug T
et al.
Regulation of K+ flow by a ring of negative charges in the outer pore of BKCa channels. Part I: Aspartate 292 modulates K+ conduction by external surface charge effect.
J. Gen. Physiol.,
2004
Aug
, 124 (173-84).
Papazian DM
et al.
Alteration of voltage-dependence of Shaker potassium channel by mutations in the S4 sequence.
Nature,
1991
Jan
24
, 349 (305-10).
Liman ER
et al.
Voltage-sensing residues in the S4 region of a mammalian K+ channel.
Nature,
1991
Oct
24
, 353 (752-6).
Xu J
et al.
Assembly of voltage-gated potassium channels. Conserved hydrophilic motifs determine subfamily-specific interactions between the alpha-subunits.
J. Biol. Chem.,
1995
Oct
20
, 270 (24761-8).
Brahmajothi MV
et al.
In situ hybridization reveals extensive diversity of K+ channel mRNA in isolated ferret cardiac myocytes.
Circ. Res.,
1996
Jun
, 78 (1083-9).
Contributors: Rajnish Ranjan, Michael Schartner, Katherine Johnston
To cite this page: [Contributors] Channelpedia https://channelpedia.epfl.ch/wikipages/19/ , accessed on 2026 May 27